生态环境学报 ›› 2026, Vol. 35 ›› Issue (5): 784-792.DOI: 10.16258/j.cnki.1674-5906.2026.05.011
李敏1,*( ), 罗嘉庆1, 周柏丰1, 王芸1, 蔺红萍1, 王斌2, 孙玉林1,3, 梁波4,*(
), 罗嘉庆1, 周柏丰1, 王芸1, 蔺红萍1, 王斌2, 孙玉林1,3, 梁波4,*( )
)
收稿日期:2025-06-10
修回日期:2026-01-09
接受日期:2026-02-15
出版日期:2026-05-18
发布日期:2026-05-08
通讯作者:
*E-mail: 作者简介:李敏(1978年生),女,副教授,博士,主要研究方向为环境微生物技术。E-mail: limin_ecit@163.com
基金资助:
LI Min1,*( ), LUO Jiaqing1, ZHOU Baifeng1, WANG Yun1, Lin Hongping1, WANG Bin2, SUN Yulin1,3, LIANG Bo4,*(
), LUO Jiaqing1, ZHOU Baifeng1, WANG Yun1, Lin Hongping1, WANG Bin2, SUN Yulin1,3, LIANG Bo4,*( )
)
Received:2025-06-10
Revised:2026-01-09
Accepted:2026-02-15
Online:2026-05-18
Published:2026-05-08
摘要:
伴随核能应用的持续推进,铀资源供应紧张与含铀废水环境风险问题逐渐显现。利用细胞表面展示技术,将人源金属硫蛋白基因表达于酿酒酵母(Saccharomyces cerevisiae)表面,通过海藻酸钠包埋并添加Fe3O4磁性颗粒,制备酵母工程菌磁性微球,研究微球对含铀废水的吸附效果;采用扫描电子显微镜(SEM)结合能谱(EDS)分析,表征磁性微球吸附U(VI)前后的表面形貌及元素分布变化。结果表明,酵母工程菌磁性微球具备良好的磁响应性(饱和磁化强度为0.62 emu·g−1),可实现水溶液中的快速磁分离;其对U(VI)的最佳吸附pH值为5.82,饱和吸附量达74.61 mg·g−1;吸附过程符合准二级动力学和Langmuir等温模型,表明其为单分子层化学吸附,并以表面官能团与U(VI)的配位作用为主导机制;热力学分析证实该过程为自发的吸热反应,升温有利于吸附进行;解吸实验显示,Na2CO3对U(VI)的解吸效率最佳(63.27%);SEM-EDS分析进一步证实,吸附后微球表面的酵母细胞有显著的颗粒物沉积并检测到U(VI)特征信号,明确了工程酵母细胞是吸附U(VI)的主要活性位点。该研究为含铀废水的治理与资源回收提供了新的功能材料与技术借鉴。
中图分类号:
李敏, 罗嘉庆, 周柏丰, 王芸, 蔺红萍, 王斌, 孙玉林, 梁波. 酵母工程菌磁性微球对水溶液中U(VI)的吸附[J]. 生态环境学报, 2026, 35(5): 784-792.
LI Min, LUO Jiaqing, ZHOU Baifeng, WANG Yun, Lin Hongping, WANG Bin, SUN Yulin, LIANG Bo. Adsorption of U(VI) from Aqueous Solution by Engineered Yeast Magnetic Microspheres[J]. Ecology and Environmental Sciences, 2026, 35(5): 784-792.
| 吸附材料 | qe, exp/(mg·g−1) | 准一级动力学模型 | 准二级动力学模型 | |||||
|---|---|---|---|---|---|---|---|---|
| q1, cal/(mg·g−1) | k1/(min−1) | R | q2, cal/(mg·g−1) | k2/(g·mg−1·min−1) | R | |||
| 磁性微球 | 69.38 | 13.91 | 7.51×10−3 | 0.9126 | 69.44 | 2.14×10−3 | 0.9983 | |
表1 微球吸附U(VI)的动力学模型参数
Table 1 Kinetics parameters for U(VI) adsorption on magnetic microspheres
| 吸附材料 | qe, exp/(mg·g−1) | 准一级动力学模型 | 准二级动力学模型 | |||||
|---|---|---|---|---|---|---|---|---|
| q1, cal/(mg·g−1) | k1/(min−1) | R | q2, cal/(mg·g−1) | k2/(g·mg−1·min−1) | R | |||
| 磁性微球 | 69.38 | 13.91 | 7.51×10−3 | 0.9126 | 69.44 | 2.14×10−3 | 0.9983 | |
| 吸附材料 | Langmuir吸附等温模型 | Freundlich吸附等温模型 | |||||
|---|---|---|---|---|---|---|---|
| KL | qm/(mg·g−1) | R | KF | n | R | ||
| 磁性微球 | 0.18 | 111.11 | 0.9711 | 25.60 | 3.00 | 0.6165 | |
表2 微球吸附U(VI)的等温模型拟合参数
Table 2 Adsorption isotherm parameters for U(VI) adsorption on magnetic microspheres
| 吸附材料 | Langmuir吸附等温模型 | Freundlich吸附等温模型 | |||||
|---|---|---|---|---|---|---|---|
| KL | qm/(mg·g−1) | R | KF | n | R | ||
| 磁性微球 | 0.18 | 111.11 | 0.9711 | 25.60 | 3.00 | 0.6165 | |
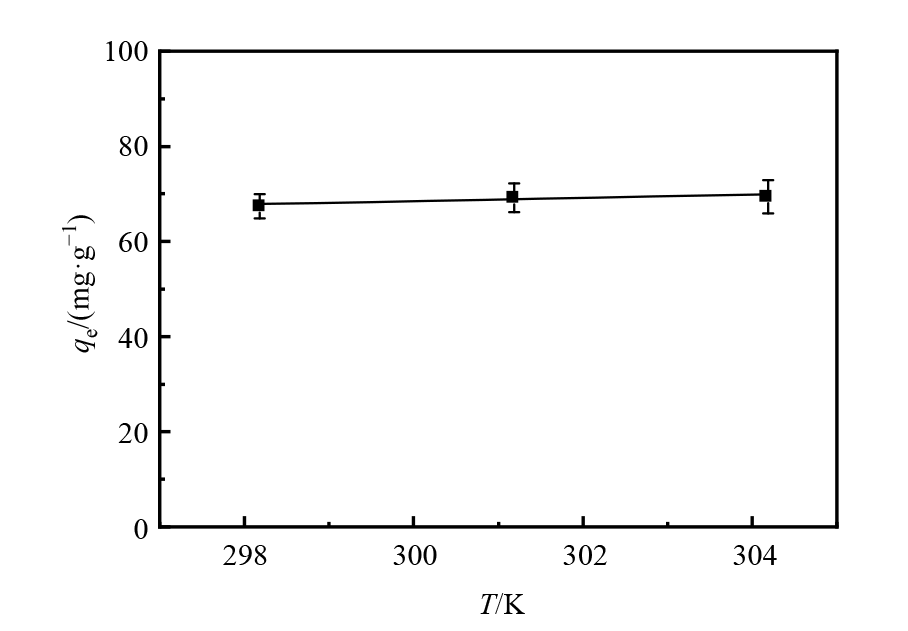
图10 温度对微球吸附U(VI)的影响
Figure 10 Effect of solution temperatures on the adsorption of U(VI) by magnetic microspheres pH=5.82;V=50 mL;m=0.03 g;C0=50 mg·L?1
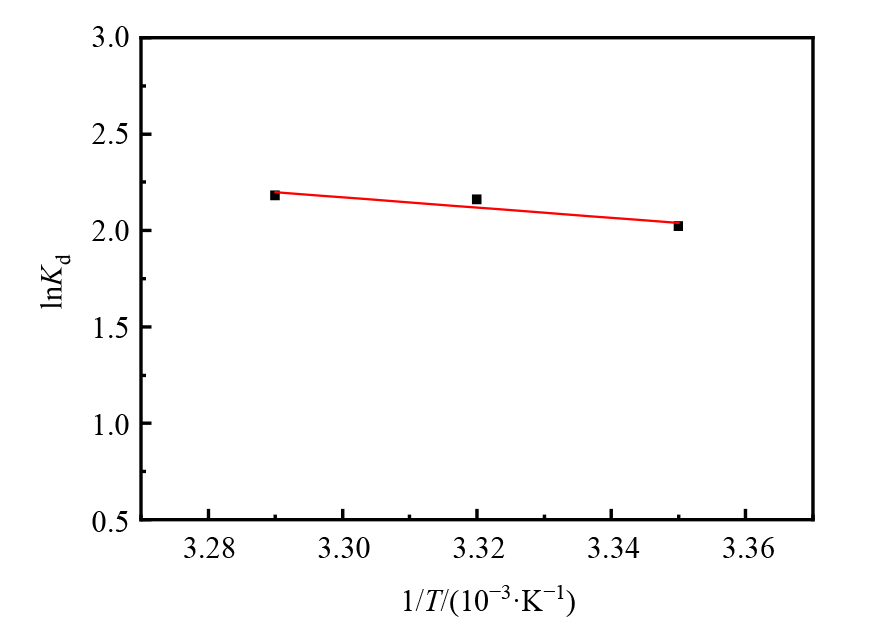
图11 微球吸附U(VI)过程中lnKd与1/T的线性相关性分析
Figure 11 Adsorption thermodynamics of U(VI) adsorption on magnetic microspheres pH=5.82;V=50 mL;m=0.03 g;C0=50 mg·L?1
| 吸附材料 | ΔH/ (kJ·mol−1) | ΔS/ (J·mol−1·K−1) | ΔG/(kJ·mol−1) | ||
|---|---|---|---|---|---|
| 298.15 (K) | 301.15 (K) | 304.15 (K) | |||
| 磁性微球 | 20.76 | 86.57 | −5.05 | −5.31 | −5.57 |
表3 微球吸附U(VI)的热力学参数
Table 3 Adsorption thermodynamic parameters for U(VI) adsorption on magnetic microspheres
| 吸附材料 | ΔH/ (kJ·mol−1) | ΔS/ (J·mol−1·K−1) | ΔG/(kJ·mol−1) | ||
|---|---|---|---|---|---|
| 298.15 (K) | 301.15 (K) | 304.15 (K) | |||
| 磁性微球 | 20.76 | 86.57 | −5.05 | −5.31 | −5.57 |
| [1] |
ABOZAID M, RABIE A, Mohamed S, 2023. Recovery of uranium from solutions using Aspergillus nidulans isolated from monazite mineral[J]. International Journal of Environmental and Analytical Chemistry, 103(17): 5422-5452.
DOI URL |
| [2] |
AKASH S, SIVAPRAKASH B, RAJA V V, et al., 2022. Remediation techniques for uranium removal from polluted environment-Review on methods, mechanism and toxicology[J]. Environmental Pollution, 302: 119068.
DOI URL |
| [3] |
BALARAM V, RANI A, RATHORE D P S, 2022. Uranium in groundwater in parts of India and world: A comprehensive review of sources, impact to the environment and human health, analytical techniques, and mitigation technologies[J]. Geosystems and Geoenvironment, 1(2): 100043.
DOI URL |
| [4] | BANALA U, DAS N, TOLETI S, 2021. Microbial interactions with uranium: Towards an effective bioremediation approach[J]. Environmental Technology & Innovation, 21: 101254. |
| [5] |
BANERJEE S, KUNDU A, DHAK P, 2022. Bioremediation of uranium from waste effluents using novel biosorbents: A review[J]. Journal of Radioanalytical and Nuclear Chemistry, 331(6): 2409-2435.
DOI |
| [6] |
BJØRKLUND G, SEMENOVA Y, PIVINA L, et al., 2020. Uranium in drinking water: A public health threat[J]. Archives of Toxicology, 94(5): 1551-1560.
DOI PMID |
| [7] |
CHEN C, HU J, WANG J L, 2020a. Biosorption of uranium by immobilized Saccharomyces cerevisiae[J]. Journal of Environmental Radioactivity, 213: 106158.
DOI URL |
| [8] |
CHEN C, HU J, WANG J L, 2020b. Uranium biosorption by immobilized active yeast cells entrapped in calcium-alginate-PVA-GO-crosslinked gel beads[J]. Radiochimica Acta, 108(4): 273-286.
DOI URL |
| [9] |
CHEN X X, CAI S H, ZHANG N, et al., 2023. Biosorption of U(VI) and mechanisms by live and dead cells of Sphingopyxis sp. YF1[J]. Environmental Science and Pollution Research, 30(50): 109469-109480.
DOI |
| [10] |
CHWASTOWSKI J, STAROŃ P, 2022. Influence of Saccharomyces cerevisiae yeast cells immobilized on Cocos nucifera fibers for the adsorption of Pb(II) ions[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 632: 127735.
DOI URL |
| [11] |
DAVENPORT B, HALLAM S J, 2023. Emerging enzyme surface display systems for waste resource recovery[J]. Environmental Microbiology, 25(2): 241-249.
DOI URL |
| [12] |
DINIS M L, FIÚZA A, 2021. Mitigation of uranium mining impacts-a review on groundwater remediation technologies[J]. Geosciences, 11(6): 250.
DOI URL |
| [13] |
GANDHI T P, SAMPATH P V, MALIYEKKAL S M, 2022. A critical review of uranium contamination in groundwater: Treatment and sludge disposal[J]. Science of the Total Environment, 825: 153947.
DOI URL |
| [14] |
GIETZ R D, SCHIESTL R H, 2007. High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method[J]. Nature Protocols, 2(1): 31-34.
DOI PMID |
| [15] |
HOSSEINI M, SOUDI M R, TAJER-MOHAMMAD-GHAZVINI P, et al., 2023. Uranium biosorption by autolyzed residues of baker yeast: Characterization and optimization[J]. Biomass Conversion and Biorefinery, 14: 22617-22632.
DOI |
| [16] |
HUSSIEN S S, SALAH M, MUHAMMAD S S, et al., 2023. Environmental bioremediation for uranium removing from aqueous solution by modified polyvinyl alcohol (PVA) embedded with Aspergillus versicolor[J]. Bioremediation Journal, 27(2): 104-115.
DOI URL |
| [17] |
JIANG Y T, YANG F, DAI M, et al., 2022. Application of microbial immobilization technology for remediation of Cr(VI) contamination: A review[J]. Chemosphere, 286(Part 2): 131721.
DOI URL |
| [18] |
JUN B M, LEE H K, PARK S, et al., 2021. Purification of uranium-contaminated radioactive water by adsorption: A review on adsorbent materials[J]. Separation and Purification Technology, 278: 119675.
DOI URL |
| [19] | KOLHE N, ZINJARDE S, ACHARYA C, 2020. Removal of uranium by immobilized biomass of a tropical marine yeast Yarrowia lipolytica[J]. Journal of Environmental Radioactivity, 223-224: 106419. |
| [20] |
LIMOUSIN G, GAUDET J P, CHARLET L, et al., 2007. Sorption isotherms: A review on physical bases, modeling and measurement[J]. Applied Geochemistry, 22(2): 249-275.
DOI URL |
| [21] | LIU M R, LI Z N, CHEN Z J, et al., 2022. Simultaneous biodetection and bioremediation of Cu2+ from industrial wastewater by bacterial cell surface display system[J]. International Biodeterioration & Biodegradation, 173: 105467. |
| [22] |
LU C W, HO H C, YAO C L, et al., 2023. Bioremediation potential of cadmium by recombinant Escherichia coli surface expressing metallothionein MTT5 from Tetrahymena thermophile[J]. Chemosphere, 310: 136850.
DOI URL |
| [23] |
LU X, ZHOU X J, WANG T S, 2013. Mechanism of uranium (VI) uptake by Saccharomyces cerevisiae under environmentally relevant conditions: Batch, HRTEM, and FTIR studies[J]. Journal of hazardous materials, 262: 297-303.
DOI URL |
| [24] |
MAKSOUD M A, ELGARAHY A M, FARRELL C, et al., 2020. Insight on water remediation application using magnetic nanomaterials and biosorbents[J]. Coordination Chemistry Reviews, 403: 213096.
DOI URL |
| [25] |
MASHANGOANE B F, CHIRWA E N, 2022. Cell surface display of palladium binding peptide on Saccharomyces cerevisiae EBY100 cells using the a-agglutinin anchor system developed for the biosorption of Pd(II)[J]. Minerals Engineering, 176: 107325.
DOI URL |
| [26] |
MEHROTRA T, DEV S, BANERJEE A, et al., 2021. Use of immobilized bacteria for environmental bioremediation: A review[J]. Journal of Environmental Chemical Engineering, 9(5): 105920.
DOI URL |
| [27] |
MISRA C S, SOUNDERAJAN S, APTE S K, 2021. Metal removal by metallothionein and an acid phosphatase PhoN, surface-displayed on the cells of the extremophile, Deinococcus radiodurans[J]. Journal of Hazardous Materials, 419: 126477.
DOI URL |
| [28] |
QI Z, WANG Q, WANG H, et al., 2021. Metallothionein attenuated arsenic-induced cytotoxicity: The underlying mechanism reflected by metabolomics and lipidomics[J]. Journal of Agricultural and Food Chemistry, 69(18): 5372-5380.
DOI PMID |
| [29] | RAEESZADEH‐SARMAZDEH M, CHEN W, 2021. Recent advances in cell surface display technologies for directed protein evolution[J]. Protein Engineering: Tools and Applications, 20: 81-103. |
| [30] |
SMEDLEY P L, KINNIBURGH D G, 2023. Uranium in natural waters and the environment: Distribution, speciation and impact[J]. Applied Geochemistry, 148: 105534.
DOI URL |
| [31] |
TEE G T, GOK X Y, YONG W F, 2022. Adsorption of pollutants in wastewater via biosorbents, nanoparticles and magnetic biosorbents: A review[J]. Environmental Research, 212(Part B): 113248.
DOI URL |
| [32] |
WANG S, RAN Y H, LU B H, et al., 2020. A review of uranium-induced reproductive toxicity[J]. Biological Trace Element Research, 196: 204-213.
DOI PMID |
| [33] |
XIE Y, LIU Z Y, GENG Y T, et al., 2023. Uranium extraction from seawater: material design, emerging technologies and marine engineering[J]. Chemical Society Reviews, 52(1): 97-162.
DOI URL |
| [34] |
XU R, WANG X Y, CHEN Y Y, et al., 2023. A betaine-modified whole-cell biocatalyst with surface display of Thermomyces lanuginosus lipase for biodiesel production from Jatropha curcas oil[J]. Industrial Crops and Products, 205: 117479.
DOI URL |
| [35] |
YU Q, CUI O, 2023. Enrichment and remediation of uranium by microorganisms: A review[J]. Open Journal of Environmental Biology, 8(1): 020-038.
DOI URL |
| [36] |
ZHANG C M, CHEN H Y, ZHU Y P, et al., 2022. Saccharomyces cerevisiae cell surface display technology: Strategies for improvement and applications[J]. Frontiers in Bioengineering and Biotechnology, 10: 1056804.
DOI URL |
| [37] |
ZHENG X Y, SHEN Y H, WANG X Y, et al., 2018. Effect of pH on uranium (VI) biosorption and biomineralization by Saccharomyces cerevisiae[J]. Chemosphere, 203: 109-116.
DOI URL |
| [38] |
ZINICOVSCAIA I, YUSHIN N, GROZDOV D, et al., 2023. The remediation of dysprosium-containing effluents using cyanobacteria Spirulina platensis and yeast Saccharomyces cerevisiae[J]. Microorganisms, 11(8): 2009.
DOI URL |
| [39] | 徐文婷, 陈国梁, 屈志慧, 等, 2023. 微生物在镉污染土壤修复中的应用及其作用机理[J]. 生物工程学报, 39(7): 2612-2623. |
| XU W T, CHEN G L, QU Z H, et al., 2023. Microbial remediation of cadmium-contaminated soils and its mechanisms: A review[J]. Chinese Journal of Biotechnology, 38(9): 3301-3315. |
| [1] | 曹振宇, 涂晨, 刘颖, 韩军超, 邢倩雯, 骆永明. 趋磁细菌Magnetospirillum gryphiswaldense MSR-1对镉的生物吸附初步研究[J]. 生态环境学报, 2025, 34(1): 99-107. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
